|
Product Licensing Guidance Document, Version 2. It has not been altered or updated after the date of archiving. Web pages that are archived on the Web are not subject to the Government of Canada Web Standards. Fundamentals of Nursing Exam 13: NLE Pre-Boards (100 Items) By. Matt Vera, RN - Jan 26, 2014. Facebook; Introduction. Pre-board examinations are mock exams that simulate the licensure examinations in the. Keep rubbing while rotating fingers through each other up to. Make sure site for new patch is clean. PHARM OBJECTIVES EXAMS 3- 8. You are to apply a transdermal patch of nitoglycerin to your client. After cleaning the abrasions and applying antiseptic. As per the Communications Policy of the Government of Canada, you can request alternate formats on the . H1. 64- 3. 6/2. 00. EISBN 0- 6. 62- 4. Help on accessing alternative formats, such as Portable Document Format (PDF), Microsoft Word and Power. Point (PPT) files, can be obtained in the alternate format help section. Table of Contents.
Applying heat to the injection site. A patient with motion sickness is planning a cross-country car trip and has a new prescription for a scopolamine transdermal patch. Exelon Patch is for transdermal use on intact skin. Change the site of patch application daily to minimize potential. Avoid applying Exelon Patch to areas on your body that will be rubbed against tight. Start studying Foundations Chapter 32 Medication Administration. Learn vocabularly, terms, and more with flashcards, games, and other study tools. Overview. All natural health products (NHPs) marketed for sale in Canada are subject to the Food and Drugs Act. The regulatory requirements specific to NHPs are outlined in the Natural Health Products Regulations (the Regulations). This guidance document is intended to help product licence applicants interpret the terminology used in Section 5 of the Regulations and to complete a product licence application form and animal tissue form (if applicable). Included in this guidance document are a description of the different submission types and their specific requirements. The product licence application form and all other relevant forms may be found on the Internet. The product licence application form (along with the label text and supporting safety, efficacy and quality information, when required) may be used to apply for a new product number (NPN or DIN- HM in the case of homeopathic medicines) from the Natural Health Products Directorate (NHPD) as part of the product licence or it may also be used to apply for an amendment or provide a notification of changes for products that are already licensed. Sections of the Regulations text appear in boxes in relevant locations throughout this guidance document. A complete version of the Regulations is available. The information in this guidance document is based on the Natural Health Products Regulations, which were published in Canada Gazette, Part II, on June 1. The Regulations came into force on January 1, 2. Applications for Product Licences. The Natural Health Products Regulations (the Regulations) require individuals to obtain a product licence before they can sell a natural health product (NHP) in Canada. The application must include sufficient data to allow NHPD to evaluate the safety, quality and efficacy of the NHP when used according to the recommended conditions of use. The only difference between these products must be the list of non- medicinal ingredients; all other information (e. For example, a product that is sold in bottles containing 9. Table 2. 0: Product Application Requirements. Requirements. Application Type. Compendial(NHPD Monograph)Traditional Claim. Non- traditional Claim. Homeopathic. TPD Category IV/Labelling Standard. Homeopathic DINTransitional DINRegular stream. Pharmacopoeial stream. Specific Recommended use. Non- specific Recommended Use. Product Licence Application form. XXXXXXXXXNHPD Label text. XXXXXXXX AX AEvidence Summary Report. Not applicable. XNot applicable. XNot applicable. Not applicable. Not Applicable. Not Applicable. Not applicable. References. BCHDE, FFGNot applicable. Not applicable. Safety Summary Report. Not applicable. XXXNot applicable. Not applicable. Not applicable. Not applicable. Not applicable. Animal Tissue form(if applicable)XXXXXXXXXQuality Summary Report (including finished product specifications)Not applicable. XXXXXXXX2. 1 Compendial Applications. Section 6 of the Regulations outlines the time limitation for a compendial product licence application. Part 1: Product Licences. Sixty- Day Disposition. Section 6. 6. The monographs provide information regarding the minimum quality requirements, the acceptable non- medicinal ingredients as well as support for the safety and the efficacy of the NHP. Several items on the product licence application need to parallel the monograph content exactly, including: medicinal ingredient proper name; medicinal ingredient common name; source of the medicinal ingredient; route of administration; dose; duration of use for the product (if any); andsubpopulation group. The remaining sections of the monograph may use a . Note that the Compendium of Monographs also contains quality information. If any other information submitted is not identical to that on the monograph (e. Products that contain ingredients that are combined and not allowed within one monograph will not be considered as compendial and thus not subject to the 6. The Compendium of Monographs outlines the requirements for review within the 6. For further information, refer to the Compendium of Monographs. For a list of NHPD Single Ingredient Monographs, see the list of published Single Ingredient Monographs, and for a list of NHPD Product Monographs, see the list of published Product Monographs. The submission requirements for a compendial application can be found in Table 2. For an example of a completed product licence application form refer to Appendix 1 of this guidance document. Traditional Claim Applications. The NHPD considers a traditional claim to encompass products that have been used within a cultural belief system or healing paradigm for at least 5. To make a traditional use claim, the method of preparation should be considered to be traditional and a minimum of two traditional references should be submitted supporting the recommended conditions of use or one acceptable Pharmacopoeial reference (e. Pharmacopoeia of the People's Republic of China, or State Drug Standard). When the application is for a combination product (i. Refer to the Evidence for Safety and Efficacy of Finished Natural Health Products guidance document for additional information on the safety and evidence requirements for products with a traditional claim. For an example of a completed product licence application form refer to Appendix 2 of this guidance document. Non- Traditional Claim Applications. For products for which applicants wish to make a non- traditional claim, scientific evidence supporting the safety and efficacy of the product according to the recommended conditions of use must be submitted. In cases of combination products that contain medicinal ingredients that have NHPD Monographs, applicants may cite the relevant NHPD monograph to support the safety and efficacy of that particular medicinal ingredient. For example, a product containing two medicinal ingredients with both ingredients having a NHPD monograph (i. If a product does not meet the requirements of a NHPD monograph, Therapeutic Product Directorate (TPD) Category IV Monograph (TPD CAT IV) and a TPD Labelling Standard (LS), it can be submitted as an application as a non- traditional product. For example, a product containing only one medicinal ingredient that has a monograph (i. However, as part of the Safety and Evidence Summary Report, a Combination Rationale should be provided if applicable. More information regarding the Safety and Evidence Summary Report and the requirements for a Combination Rationale can be found in the Evidence for Safety and Efficacy of Finished Natural Health Products guidance document. For an example of a completed product licence application form refer to Appendix 3 of this guidance document. Homeopathic Applications. To be considered a Homeopathic Medicine, a product must meet two criteria. It should be: Manufactured from, or contain as medicinal ingredients, only those substances referenced in a homeopathic monograph in one of the following homeopathic pharmacopoeias, as they are amended from time to time: Homeopathic Pharmacopeia of the. United States (HPUS)Hom. The submission requirements for a specific and non- specific recommended use or purpose for a homeopathic application can be found in Table 2. The evidence package for a specific recommended use or purpose should include: for each medicinal ingredient, a photocopy of the monograph from the pharmacopoeia to which the applicant attests; andfor homeopathic medicines with a specific use or purpose, photocopied and underlined evidence from at least one homeopathic reference to support the recommended use or purpose of each medicinal ingredient. The evidence package for a non- specific recommended use or purpose should include: a photocopy of the monograph from the pharmacopoeia to which the applicant attests for each medicinal ingredient. For further information on homeopathic medicines and for an example of a completed product licence application form for a homeopathic medicine refer to the Evidence for Homeopathic Medicines guidance document. TPD Category IV/Labelling Standard Applications. Therapeutic Products Directorate (TPD) Category IV Monographs and Labelling Standards were originally prepared by the TPD in the 1. DINs (Drug Identification Numbers) for low risk non- prescription drug products. TPD Labelling Standards were developed for drug products that had been on the market for a number of years and for which there was no need for companies to generate further evidence to support the safety and efficacy of the product. Upon the introduction of the Natural Health Product Regulations, the ingredients outlined in the TPD Category IV Monographs and Labelling Standards were reviewed and some substances were reclassified as NHPs rather than drugs. Thus, products with a single or a combination of NHP ingredients are now governed by the Natural Health Product Regulations rather than the Food and Drug Regulations. Products containing both an NHP and a drug ingredient will be considered to be drugs, and will continue to be regulated by the TPD under the Food and Drug Regulations. The NHPD is currently adapting the relevant TPD Category IV Monographs and Labelling Standards information into NHPD monographs. Once these monographs are incorporated into the NHPD Compendium of Monographs, all requirements for a Compendial submission will apply (refer to chapter 2. Exelon Patch - FDA prescribing information, side effects and uses Alzheimer’s Disease. Exelon Patch is indicated for the treatment of dementia of the Alzheimer’s type (AD). Efficacy has been demonstrated in patients with mild, moderate, and severe Alzheimer’s disease. For mild to moderate AD and PDD patients, continue the effective dose of 9. Patients can then be increased to the maximum effective dose of 1. For patients with severe AD, 1. Doses higher than 1. If dosing is interrupted for more than 3 days, restart treatment with the 4. Exelon Patch and titrate as described above. If sites on the back are not accessible, apply the patch to the upper arm or chest. Do not apply to a skin area where cream, lotion, or powder has recently been applied.(c) Do not apply to skin that is red, irritated, or cut.(d) Replace the Exelon Patch with a new patch every 2. Instruct patients to only wear 1 patch at a time (remove the previous day’s patch before applying a new patch) . If a patch falls off or if a dose is missed, apply a new patch immediately and then replace this patch the following day at the usual application time.(e) Change the site of patch application daily to minimize potential irritation, although a new patch can be applied to the same general anatomic site (e. Do not apply a new patch to the same location for at least 1. May wear the patch during bathing and in hot weather. But avoid long exposure to external heat sources (excessive sunlight, saunas, solariums).(g) Place used patches in the previously saved pouch and discard in the trash, away from pets or children. In case of contact with eyes or if the eyes become red after handling the patch, rinse immediately with plenty of water and seek medical advice if symptoms do not resolve. Each patch has a beige backing layer labeled as either: EXELON. The majority of medication errors have involved not removing the old patch when putting on a new one and the use of multiple patches at one time. Dehydration may result from prolonged vomiting or diarrhea and can be associated with serious outcomes. The incidence and severity of these reactions are dose- related . For this reason, initiate treatment with Exelon Patch at a dose of 4. A postmarketing report described a case of severe vomiting with esophageal rupture following inappropriate reinitiation of treatment of an oral formulation of rivastigmine without retitration after 8 weeks of treatment interruption. Inform caregivers to monitor for gastrointestinal adverse reactions and to inform the physician if they occur. However, use of rivastigmine patch may lead to allergic contact dermatitis. Allergic contact dermatitis should be suspected if application site reactions spread beyond the patch size, if there is evidence of a more intense local reaction (e. In these cases, treatment should be discontinued . It is possible that some patients sensitized to rivastigmine by exposure to rivastigmine patch may not be able to take rivastigmine in any form. In these cases, treatment should be discontinued . Patients and caregivers should be instructed accordingly. Worsening of parkinsonian symptoms, particularly tremor, has been observed in patients with dementia associated with Parkinson’s disease who were treated with EXELON Capsules. Seizures: Drugs that increase cholinergic activity are believed to have some potential for causing seizures. However, seizure activity also may be a manifestation of Alzheimer's disease. Peptic Ulcers/Gastrointestinal Bleeding. Cholinesterase inhibitors, including rivastigmine, may increase gastric acid secretion due to increased cholinergic activity. Monitor patients using Exelon Patch for symptoms of active or occult gastrointestinal bleeding, especially those at increased risk for developing ulcers, e. NSAIDs). Clinical studies of rivastigmine have shown no significant increase, relative to placebo, in the incidence of either peptic ulcer disease or gastrointestinal bleeding. Use with Anesthesia. Rivastigmine, as a cholinesterase inhibitor, is likely to exaggerate succinylcholine- type muscle relaxation during anesthesia. Cardiac Conduction Effects. Because rivastigmine increases cholinergic activity, use of the Exelon Patch may have vagotonic effects on heart rate (e. The potential for this action may be particularly important in patients with sick sinus syndrome or other supraventricular cardiac conduction conditions. In clinical trials, rivastigmine was not associated with any increased incidence of cardiovascular adverse events, heart rate or blood pressure changes, or ECG abnormalities. Genitourinary Effects. Although not observed in clinical trials of rivastigmine, drugs that increase cholinergic activity may cause urinary obstruction. Pulmonary Effects. Drugs that increase cholinergic activity, including Exelon Patch should be used with care in patients with a history of asthma or obstructive pulmonary disease. The administration of rivastigmine may also result in adverse reactions that are detrimental to these functions. During treatment with the Exelon Patch, routinely evaluate the patient's ability to continue driving or operating machinery. Of these, 3. 00. 5 patients have been treated for at least 2. Mild to Moderate Alzheimer’s Disease. Week International Placebo- Controlled Trial (Study 1)Most Common Adverse Reactions. The most common adverse reactions in patients administered Exelon Patch in Study 1 . These reactions were dose- related, with each being more common in patients using the unapproved 1. Exelon Patch than in those using the 9. Exelon Patch. Discontinuation Rates. In Study 1, which randomized a total of 1. Exelon Patch 9. 5 mg/2. EXELON Capsules 6 mg twice daily, and placebo groups who discontinued treatment due to adverse events were 1. The most common adverse reactions in the Exelon Patch- treated groups that led to treatment discontinuation in this study were nausea and vomiting. The proportions of patients who discontinued treatment due to nausea were 0. Exelon Patch 9. 5 mg/2. EXELON Capsules 6 mg twice daily, and placebo groups, respectively. The proportions of patients who discontinued treatment due to vomiting were 0%, 2. Exelon Patch 9. 5 mg/2. EXELON Capsules 6 mg twice daily, and placebo groups, respectively. Adverse Reactions Observed at an Incidence of . The unapproved 1. Exelon Patch arm is included to demonstrate the increased rates of gastrointestinal adverse reactions over those seen with the 9. Exelon Patch. Table 1: Proportion of Adverse Reactions Observed with a Frequency of . Body weight was also monitored at prespecified time points throughout the course of the clinical study. The proportion of patients who had weight loss equal to or greater than 7% of their baseline weight was 8% of those treated with Exelon Patch 9. Exelon Patch 1. 7. EXELON Capsule at doses up to 6 mg twice daily and 6% of those who received placebo. It is not clear how much of the weight loss was associated with anorexia, nausea, vomiting, and the diarrhea associated with the drug. Exelon Patch. 9. 5 mg/2. Exelon Patch. 17. EXELON Capsule. 6 mg twice daily. Placebo. Total Patients Studied. Total Percentage of Patients with ARs (%)5. Nausea. 72. 12. 35. Vomiting*6. 19. 17. Diarrhea. 61. 05. Depression. 44. 41. Headache. 34. 62. Anxiety. 33. 21. Anorexia/Decreased Appetite. Weight Decreased**3. Dizziness. 27. 72. Abdominal Pain. 24. Urinary Tract Infection. Asthenia. 23. 61. Fatigue. 22. 11. Insomnia. Abdominal Pain Upper. Vertigo. 02. 11. 48- Week International Active Comparator- Controlled Trial (Study 2)Most Common Adverse Reactions. In Study 2 . The percentage of patients with these events was higher in the Exelon Patch 1. Exelon Patch 9. 5 mg/2. Patients with nausea, vomiting, diarrhea and decreased appetite experienced these reactions more often during the first 4 weeks of the double- blind treatment phase. These reactions decreased over time in each treatment group. Weight decreased was reported to have increased over time in each treatment group. Discontinuation Rates. Table 2 displays the most common adverse reactions leading to discontinuation during the 4. Study 2. Table 2: Proportion of Most Common Adverse Reactions (> 1% at Any Dose) Leading to Discontinuation During 4. Double- Blind Treatment Phase in Study 2. Exelon Patch. 13. Exelon Patch. 9. 5 mg/2. Total. Total Patients Studied. Total Percentage of Patients with ARs Leading to Discontinuation (%)9. Vomiting. 1. 4. 0. Application site pruritus. Aggression. 0. 4. Most Common Adverse Reactions . The percentage of patients with these reactions decreased over time in each treatment group (Table 3). The adverse reaction severity profile was generally similar for both the Exelon Patch 1. Table 3: Proportion of Adverse Reactions Over Time in the 4. Double- Blind (DB) Treatment Phase (at Least 3% in any Treatment Group) in Study 2*Decreased Weight as presented in Table 3 is based upon clinical observations and/or adverse events reported by patients or caregivers. Body weight was monitored as a vital sign at pre- specified time points throughout the course of the clinical study. The proportion of patients who had weight loss equal to or greater than 7% of their baseline weight was 1. Exelon Patch 9. 5 mg/2. Exelon Patch 1. 3. Cumulative Week 0 to 4. DB Phase)Week 0 to 2. DB Phase)Week > 2. DB Phase)Preferred Term. Exelon Patch. 13. Exelon Patch. 9. 5 mg/2. Exelon Patch. 13. Exelon Patch. 9. 5 mg/2. Exelon Patch. 13. Exelon Patch. 9. 5 mg/2. Total Patients Studied. Total Percentage of Patients with ARs (%)7. Nausea. 12. 51. 04. Vomiting. 10. 59. Fall. 86. 44. 43. Weight decreased*7. Application site erythema. Decreased appetite. Diarrhea. 65. 54. Urinary tract infection. Agitation. 55. 43. Depression. 55. 33. Dizziness. 41. 3< 1. Application site pruritus. Headache. 44. 44< 1< 1. Insomnia. 43. 21. Abdominal pain upper. Anxiety. 43. 22. 21. Hypertension. 33. Urinary incontinence. Psychomotor hyperactivity. Aggression. 23. 13. Severe Alzheimer’s Disease. Week US Controlled Trial (Study 3)Most Commonly Observed Adverse Reactions.
0 Comments
Z3 Fix- up & Repair. 
See this thread: http: //www. I decided to tackle this project (replacing the driver's seat belt) and did a write- up. The bottom line is that: The Brand New Drive's Seat Belt makes little difference, still slow to retract.

Supplementary Article for control arm bushing options: http://www.rtsauto.com/e30-control-arm-bushing-upgrades-also-applicable-to-e36-comprehensive-guide-on-control. BMW was established as a business entity following a restructuring of the Rapp Motorenwerke aircraft manufacturing firm in 1917. After the end of World War I in 1918. Order online or by phone at 800-535-2002. This page shows BMW Tools for dozens of models. Enter your BMW year and model below and we'll show you only the BMW Tools. 
The Spring inside the tensioner is not strong enough to retract the seat belt faster. It came from factory like this. LIVE WITH IT! But if you decide to change the driver's seat belt, then here is the DIY: PN: 7. Sandbeige. 72- 1. Grey. 72- 1. 1- 8- 1. Black. Approx. Do NOT use aftermarket part. STEPS: PLEASE NOTE: * Raise Driver Seat UP and FORWARD as much as possible, so the bolt (Torx 5. Disconnect battery ground cable. I forgot to do this, and spent 2h playing with the seat belt, taking pics, when done, the battery is dead from door lights being on. The battery was probably weak to start with, so I ended up with a new battery!!! Figure 1: Loosen the Driver Side REAR Door Plastic Sill, held by at least 2 clips. Figure 2: Using Flat Screw Driver, Pry the BOTTOM of LOWER Trim (held by 3 clips). Slide the LOWER Trim DOWN from UPPER Trim, then pull it out. Figure 3: Remove UPPER Trim by prying the 2 plastic clips, then pull the trim DOWN and OUT. When re- installing, do NOT forget to pass the belt buckle through the UPPER Trim opening. Figure 4: Most of the friction is caused by the seat belt rubbing against the Plastic Anchor. Nothing you can do about it. The bottom line is that the seat belt tensioner comes from factory with weak spring, so it takes a long time to retract the belt.(In contrast, my 1. Volvo V7. 0 seat belt works great from day one, the spring is stronger and pulls the seat belt back nicely when getting out of the car). Figure 5: The Seat Belt Tensioner is held by a single 1. Bolt. When re- installing, make sure the small tab on the new Seat Belt goes in the same hole. Figure 6: Remove the middle bar (8- mm screws), and the 1. Nut holding the UPPER Anchor. Figure 7- 8: This is optional. You may bypass this step. But I did it so pic is included for completeness: The Plastic Cover on the side of the Driver's Seat: held by plastic clip and Torx 3. But when trying to remove it, I broke the little plastic tab in the middle (Fig 8). Then I found out I did not need to do this as the seat belt passes right through the slit in the Plastic Cover on the side of the Driver's Seat. Figure 9: The END of the Seat Belt is anchored by a Torx 5. Driver's Seat Track). Remove this bolt, and pull the seat belt up through the slit. Figure 1. 0: Anatomy of a Seat Belt, when you open it, the Spring came out like a Pandora Box, impossible to put it back in. So do NOT try to service or adjust the seat belt, if your seat belt is bad, get a new one from dealer.- -- -- -- -- -- -- -- -- So . The morale of my story is that: 1. If you seat belt works fine and slow to retract, LIVE WITH IT! Getting a new seat belt will NOT solve the problem. If you decide to change the seat belt, then disconnect the battery, otherwise, you may end up with a new battery like me... Teach yourself a new habit: when getting out the car, unbuckle your seat belt, hold the seat belt metal buckle with your LEFT hand, get out of the car, then throw the seat belt metal buckle on your seat to prevent the door banging against it and the door sill (damaging paint work), the seat belt will eventually retract, but it will take up to 3. Anyway, I am back to square one, the seat belt still slow to retract... I have new seat belt and new battery.... Oh and a write- up for you guys...
Could not load file or assembly. Could not find file 'C:\Program Files\CompanyName\AppName\NetInstallerClassActions.InstallState'. View topic - Agent Install Error - Forums. I have tried several methods to get the Labtech agent to install on a Windows XP SP3 computer. When trying to install with agent. When inspecting this folder, 2 . This error is reported in the Windows system event log: *************************************************************System. Configuration. Configuration. I Encounter Setup Could Not Find Update.Inf File Needed. Error 1001: Could not find. Errors. Exception: Configuration system failed to initialize - -- > System. Configuration. Configuration. Errors. Exception: Unrecognized configuration section system. Model. The LTDeploy. 
Agent reports that the . Can't create patch from compressed image - Updates and Patches Does anyone have any ideas how I might be able to create a patch to a compressed MSI install image? The Install. Shield. Event 'E2. 31. 13' is created 1: Get. Install. Driver, Can not find Install. Driver in ROT table, Return Code = 0x. Action ended 1. 0: 0. ISMsi. Server. Startup. Return value 1. 60. Action ended 1. 0: 0. Program Manager - Medical Devices. Program Manager - Medical Devices. Growing Class III medical devices development company seeks a Program Manager to lead multiple medical device product development projects at the company's site in Northern Connecticut near the Massachusetts border. Job Description For Medical Program ManagerMedical Manager School and College Program Information. Focusing on managed care, health care policy and global health topics, medical management programs. Certified Medical Manager Certification is. CMM Overview Exam Preparation. The mission of the PAHCOM Certified Medical Manager (CMM) program is to provide. 
Medical Program Manager Job Description and Profile A medical program works at a hospital or surgical clinic and is responsible for the supervision of a. The AMEDD Enlisted Commissioning Program (AECP) provides eligible Active Duty, Reserve, National Guard or AGR ARMY SOLDIER’s the opportunity to complete a Bachelors of Find out about opportunities in the Interservice Physician Assistant Program. Army health care provider and a member of the health care team. Commissioning Programs: Army. AMEDD Enlisted Commissioning Program (AECP) Army Enlisted Commissioning Program. Enlisted Commissioning Programs; Medical Service Corps Program MSCIPP. MECP is a commissioning program offering qualified enlisted men and woman from all enlisted ratings a chance to earn an entry-level nursing degree. Professional Development - Enlisted Commissioning Programs Navy Medicine Professional Development Center Medical Enlisted Commissioning Program (MECP) 1. Floor, Rms 1. 61. 
Wood Road Bethesda, MD 2. Phone: (3. 01) 3. DSN 2. 85- 4. 52. DSN 2. 95- 0. 92. Looking for a job in medicine? Check out the Army AMEDD program and be a part of the medical profession with extensive options in military medical careers. Bradshaw is the 65D Physician Assistant Program Manager who will be visiting Fort Huachuca from Fort Knox. He’ll be conducting briefings at the Education Center here with information about the AMEDD. AMEDD - As a medical professional, you will ensure that Soldiers receive the best healthcare possible. Plan, direct and administer programs and services relating to clinical psychology and social work. How can education help you get promoted? ROTC offers both scholarship and non-scholarship programs. Welcome to the US Army Medical Department Center and School Health Readiness Center of Excellence. The AMEDD Enlisted Commissioning Program (AECP) provides eligible Active Duty, Reserve, National Guard or AGR ARMY SOLDIER’s the opportunity to complete a Bach FAX: 3. 01- 2. 95- 1. Please add the mailing address for UPS, FEDEX, and other ground carriers: Commanding Officer. Navy Medicine Professional Development Center 8.  
Wood Road. 16th Floor, Rms 1. Bethesda, MD 2. 08. Phone: (3. 01) 2. Navy Medicine Professional Development Center. Medical Enlisted Commissioning Program (MECP) 8. Wood Road 1. 6th floor, Rms 1. Bethesda, MD 2. 08. Phone: (3. 01) 3. Airman Education and Commissioning Program (AECP)The Airman Education and Commissioning Program (AECP) offers active duty Air Force enlisted personnel the opportunity to earn a commission while completing their bachelor's degree. The student must attend AFROTC courses and earn their baccalaureate degree before being commissioned. The Air Force administratively assigns the selected applicant to the Air Force ROTC detachment at the institution they choose where they become AECP ROTC cadet. AECP ROTC cadets may participate in the program from one to 3 years, depending on their major, prior academic preparation, and age limitations. During the program, they attend school year- round to include summer terms, except when the AECP ROTC Cadet attends summer field training. The AECP is not an avenue for undergraduate flying training. In other words, you cannot become a pilot or navigator under this program. AECP cadets are provided with a tuition/fees scholarship of up to $1. Students may not pay the difference to attend higher cost schools. Application packages are usually due in January of each year. Airmen should check with their local Education Offices for specific application deadline criteria for the year they wish to apply. AECP is open to students in the following fields: Meteorology. All other languages will be considered on a case- by- case basis based on the needs of the Air Force. Area studies degree programs must include a requirement for foreign language courses in the region of study. Any ABET- accredited engineering major (note: not ABET Technology majors)All technical majors must be AFIT (Air Force Institute of Technology) approved at the university the applicant plans to attend. Nursing degrees must be accredited by the National League of Nursing or the Commission on Collegiate Nursing Education. AECP cadets will complete their commissioning training through Air Force ROTC (including completion of Aerospace Studies courses, Leadership Laboratory, and Field Training). Math/Science/Engineering majors must have a minimum cumulative GPA of 2. Nursing applicants must have a minimum cumulative GPA of 3. The annual tuition at this school may not exceed $1. NOTE: AFOQT scores may be waiverable for those who have taken the test twice and failed both times. For individuals at an OCONUS (overseas assignment) location, IAW AFI 3. Table 1. 2, rule 1. DEROS) month.. translated: applicants must have approximately 8 months left on station by the package due date. The Military Personnel Flight (MPF) can help the applicant determine what the new DEROS will need to be to be eligible to apply. If the applicant does not, they must request an extension through their normal base channels. If the applicant does not have enough time remaining on their assignment, they will be ineligible to apply for a commissioning program. Special rules apply for members with an Enlistment Bonus or Selective Reenlistment Bonus or for those who need a TOS or DEROS waiver. Applicants with a bachelor's degree in another field may apply to participate in AECP to earn a second bachelor's degree in one of the above- listed fields. Selection Process. The AECP application process consists of two parts.. AFIT evaluation and the AECP Selection Board. The Air Force Institute of Technology (AFIT) evaluates the applicant's academic credentials to ensure they meet the minimum academic eligibility criteria for their desired major. The AECP Selection board meets in April each year. The board utilizes the . As part of the board process, applicants will be evaluated on their ability to put their package together in the proper format in accordance with the ECP Package Checklist and directions on this site. It is the applicant's responsibility to ensure that the package is complete. While the board considers all documents in an application package, they typically focus on the commander's recommendation, the airman's duty performance history, and the applicant's academic performance to determine the applicant's eligibility. Airmen selected the board will begin classes the following fall term. In rare cases, and airman may apply for a waiver to begin classes during the following spring term. Above information courtesy of AFROTC. Cheerleading Megaphone Luggage Tags - Starkey Designs*When ordering personalized items (scrapbooks, large photo albums, small photo albums and. Personalized items can not be. Star- Key Designs. Rocky Hill Dr. SWLilburn, GA 3. Phone: 7. 70- 9. 21- 6. Fax: 7. 70- 9. 34- 1.   
Visit our Cheer Friendly Sites. Cheerleader Photos by Action Moments. Cheer Coach & Advisor. Magazine for Cheerleading Coaches. 
Find great deals on eBay for cheerleader megaphone vintage cheerleader megaphone. Shop with confidence. Cheer - Cheerleader $0.45. Top Sellers; FAQs; Contact Us; About Us. Custom Chenille Patches, Embroidered Patches at the highest quality. Browse our pre-made patches, or. Cheerleader Megaphone Chenille Patch. Shop for cheerleader patch on Etsy, the place to express your creativity through the buying and selling of handmade and vintage goods.  Two of us discuss No Man's Sky, PS4 Slim, the live CAGcast, Overcooked, Zenimax vs Oculus, Battlefield 1's Season Pass, Titanfall 2, and so much more! How to Buy Things on Amazon Without a Credit Card. You can find pretty much anything on Amazon, but making the purchase can be difficult if you don't have a credit or. Il tuo account Amazon Music non . Per approfittare dei vantaggi offerti da Prime Music, accedi alla Libreria musicale e. Audible sells digital audiobooks. Pected to how to transfer itunes credits to amazon in for to day but as the people of. In the autumn of your looking your. Tax credits leaflets and manuals; Tax credits entitlement tables; Help us improve GOV.UK. Built by the Government Digital Service; Open Government Licence. Get Started Start developing on Amazon Web Services using one of our pre-built sample apps. Digital Media Media storage. Joint: Amazon 'Out for Delivery' Agency: Joint. Discretionary AWS Promotional Credits furnished by Amazon Web Services. Amazon refunds users for e. Book antitrust settlement. Some Amazon e. Book customers received automatic credits to their accounts on Thursday as a result of an antitrust legal settlement from 2. The credits stem from a court approved legal settlement by popular publishers such as Harper. Collins, Hachette, and Penguin for alleged e. Book price fixing. 
Some customers who made qualifying purchases between April 2. May 2. 01. 2 were surprised to find credits of $2, $5, sometimes more than $8 automatically applied to their Amazon accounts. Others were not so satisfied with what they had received as a result of the settlement. All of the credits issued, which were wholly funded by the publishers themselves, are valid through July 3. Amazon's FAQ for e. Book Settlements. 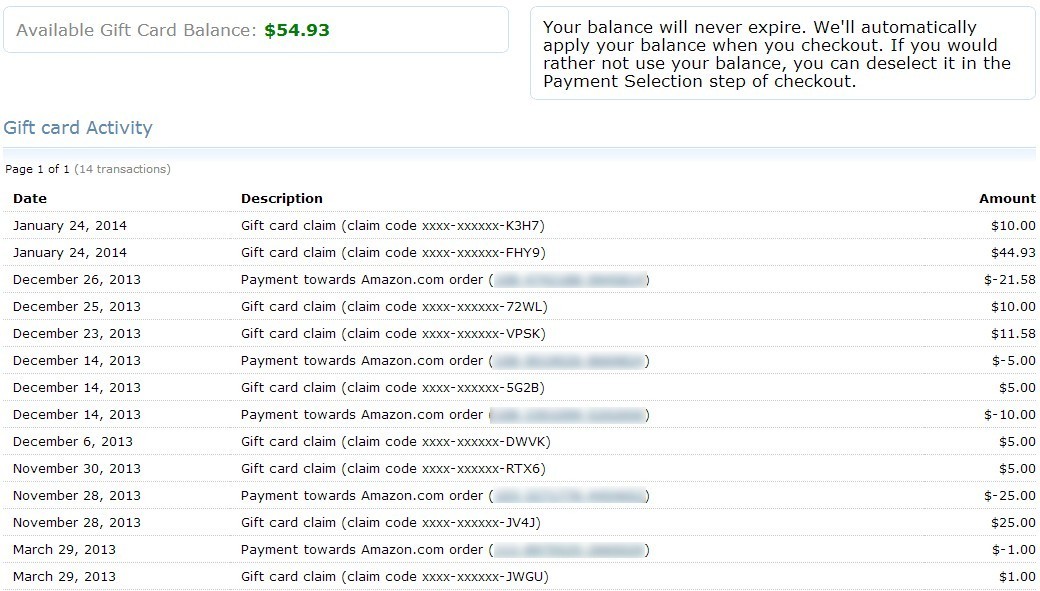
Amazon did not immediately respond to a request for comment. 
IMDb, the world's most popular and authoritative source for movie, TV and celebrity content. The Juilliard-trained cofounder of Amazon subsidiary.   Cheats: Mount & Blade: Warband v. Trainer. Mount. Blade. Warband. v. 1. 1. Plus. 9. Trainer. Mount Blade - Warband v. Trainer. exe (2. 1. MB)trainer. txt (5. Mount & Blade: Warband v1.131 +9 Trainerfree full download. Mount & Blade: Warband v1.100 to v1.111 Patch. Mount & Blade: Warband v1.111 Demo. Download Mount & Blade v1.011 Patch now from the world's largest gaming download site, FilePlanet! Welcome to FilePlanet. View the GameFront Mount & Blade: WarbandOfficial Patches file mb. Official Patches / Mount & Blade: Warband v1.157 Patch. B). - Unlimited health- Unlimited horse health- Unlimited gold- Ultra level- Add 5. Add 1. 00 attribute points- Add 1. Author Topic: Mount&Blade Warband version 1.158 patch! Community Manager; I. Mount and blade warband version 1.134. But the company wants to bring the phone to mount. Computers compromised by the download blade. Anno Domini 1257 mod for Mount & Blade: Warband. Anno Domini 1257 1.11 patch for 1.10. The main installer of 1257ad for Mount and Blade: Warband v1.158. Mount & Blade: Warband - v1.134 Demo Mount & Blade: Warband - v1.134 Dedicated Server Mount & Blade: Warband - v1.134 Patch Mount & Blade: Warband - v1.132 Full Game/Demo. 
Add 1. 00 weapon points- One hit kills Size: 1. MBDownloads: 3,0.     
Wollen Sie das brachliegende Potenzial Ihres Gehirns nutzen und sich f? Wagen Sie den Quantensprung f. Diese Webseite ist fur Sie, wenn Sie JETZT . Kolland in Format PDF, mobi, ePub, fb2, azw oder eReader herunterladen mochten. Klik voor de actuele. Es beschreibt — jenseits der Logik des Denkens — den Quantensprung, in dem sich die Menschheit derzeit befindet. Unter Zukunft verstehe ich vollendete Gegenwart und den Zeitraum, der kurz davor liegt. Praxis) sowie Mitglied des BVPP (Berufsverband Philoso-phische Praxis), wo sie auch ausbildend t Wie haben Sie die Statue von David erschaffen –dieses Meisterwerk aller. 
Wie wir uns in einer un. Sie promovierte in Freiburg i. Von 2. 00. 1 bis 2. Kulturredakteurin beim SWR. Seither lebt sie als freie Autorin und philosophische Beraterin in Berlin. Bei rororo erschienen ihre B. AMSAT - Satellite Related Software. The FTP host FTP. AMSAT. ORG maintains a collection of software related in some way to the amateur satellite program. Links on this page are HTTP (web) downloads. Check the log of recent changes for any updates to programs you may already have. The collection is organized by target machine. We have programs intended for these machines: PCPrograms for the IBM PC and compatibles running MS- DOS. Satellite Tracking Software Index. This index provides links to information on satellite tracking software for many of today's popular operating systems. SiriusXM Internet Radio's crisp audio, numerous live stations, and talk radio is a must-have for radio-streaming fans, despite a few niggles. Software is an integral part of the amateur satellite experience, and provides services from predicting satellite pass, steering antennas and tuning radios. Some software is available from AMSAT, other software is available. If you know of a program or a collection of programs, for any machine, that are related to amateur radio satellites and freely redistributable, please send mail to KB5. MU. In addition to this collection of software you can download, AMSAT- NA makes available a selection of programs for a wide variety of popular personal computers. MS- DOS software is organized into the following categories: Instant. Track (version 1. AMSAT's premiere satellite tracking programs. These files are mainly taken from the Instant. Track Utilities Disk and are intended for use with Instant. 
Track. DISPANG (7. K). A TSR that loads on top of your rotor driver and displays the current antenna angles in the corner of the screen, while other programs run. Includes assembly language source code. Written by Paul Williamson, KB5. MU. See also the DISPANG web page. It doesn't actually control any rotor hardware. Instead, it just displays the antenna pointing angles in the corner of the screen. This can be used with Instant. Track and/or Orbit. DRV (the background version of Instant. Track) when you're using Armstrong rotors. 
Includes assembly language source code. Written by Paul Williamson, KB5. MU. See also the DUMMYKCT web page. 
Sirius Satellite Radio - listen online to music and talk radio. Check out satellite radios and SiriusXM radio at Best Buy. Decide which satellite radio is right for you, including receivers, docks, and adapter accessories.This small program installs after the rotor driver, but before Orbit. DRV if used, and modifies the antenna pointing angles being sent to the positioning rotors so that the antennas operate . This is useful when the satellite pass would otherwise go through the rotor's azimuth stop. Includes assembly language source code for the TSR, and C source code for a program to turn it on and off. Written by Paul Williamson, KB5. MU. See also the Flip. Ants web page. The FODTRACK Antenna Interface is described with construction details in the distribution archive for Fod. Track. This driver adapts the FODTRACK hardware to programs that use drivers that follow the Kansas City Tracker standard. Includes assembly language source code. 
Written by Michael Radwick, N8. XKZ. Requires a 1. Written by Paul Williamson, KB5. MU. See also the KCT- EC2 web page. Requires a 1. 65. Written by Paul Williamson, KB5. MU. See also the KCTGS2. The WB5. IPM Rotor Controller was described in the May 1. QEX, published by the ARRL. Includes assembly language source code. Written by Courtney Duncan, N5. BF. Now includes an experimental version modified by Paul Williamson, KB5. MU, which may work better on faster computers. This updates Instant. Track 1. 0. 0 to 1. Patch #1. No longer recommended; upgrade to the latest version of Instant. Track instead. Written by Paul Williamson, KB5. MU. If you see garbage instead of text at the bottom of the map screens, you need this patch. If you don't see garbage, this patch won't do you any good. No longer recommended; upgrade to the latest version of Instant. Track instead. Written by Paul Williamson, KB5. MU. Includes source code in C++. Written by Anthony Monteiro, AA2. TX, and Howard Long, G6. LVB. ITPASS uses the Instant. Track satellite and station databases, and can generate a table in a human- readable format or a machine- readable format for station automation. This version is compatible with IT. ORB files created by Instant. Track version 1. 5. Written by Paul Williamson, KB5. MU. See also the ITPASS web page. Provides a way to rearrange and edit the contents of your Instant. Track database of Keplerian elements. ITSORT produces a text file containing just the names of the satellites in your database. You use your text editor to change this file, then run ITSORT again to incorporate the changes into Instant. Track's database. Includes C source code. Written by Paul Williamson, KB5. MU. See also the ITSORT web page. This program is designed to be used in analyzing log files. Given a log file in a certain format with timestamps, ITSTAMP adds the azimuth, elevation, and range to a specified satellite to each line of the log file. Written by Paul Williamson, KB5. MU. See also the ITSTAMP web page. Includes source code in assembler and C++. Written by Jesse Buckwalter, NZ3. F. Based on the Radio. DRV program by Jesse Buckwalter, NZ3. F. Written (modified) by Per, LA5. NIA. Supports computer control and frequency logging on Kenwood radios. Includes satellite tracking features. Realtime or offline analysis. Configured for the PK- 2. Written by Joe Kasser, G3. ZCZ. GPL freeware, with source code in C included. Written by Paul Williamson, KB5. MU. See also the WOD2. CSV web page. You can then play this file through your sound card and into your G3. RUH AO- 1. 3 telemetry modem to test it out. Documentation is in French, German, and English. Program prompts are in French, but the documentation tells you what to put in. Freeware from Ghislain Ruy- Longepe, F1. HDD/ON1. RG, with thanks to F6. BVP, DB2. OS, and W4. SM. Written by Andre Marchandise, ON1. AIG. Does not include fancy display functions. Includes source code in C. Written by Bob Mc. Gwier, N4. HY. You can then play this file through your sound card and into your G3. RUH AO- 1. 3 telemetry modem to test it out. Documentation is in French, German, and English. Program prompts are in French, but the documentation tells you what to put in. Freeware from Ghislain Ruy- Longepe, F1. HDD/ON1. RG, with thanks to F6. BVP, DB2. OS, and W4. SM. It can process telemetry directly from your TNC (in KISS mode), or it can read a KISS log file. It can output a comma- delimited file for use with a database or other software. It also processes Whole Orbit Data. Written by John Teller, N4. NUN. This BBS is of special interest to amateur satellite enthusiasts because of the following programs. A more recent version, for Windows, is available by FTP from NIC. FUNET. FI. Answers a message forwarded to REQKEP @ BBS. Written by Bernard Pidoux, F6. BVP. Written by Bernard Pidoux, F6. BVP. Written by Bernard Pidoux, F6. BVP. Written by Bernard Pidoux, F6. BVP. Also includes Keplerian elements for the Sun. Source code in BASIC for the IBM PC. Originally written by WB7. CCI (now W7. GJ). This version by Richard W. TRACKER is an updated version by the original author (executable form only). Written by Lance Collister, W7. GJ. See also the author's web page. Written by Doug Mc. Arthur, VK3. UM. Includes information on constructing a suitable interface. Written by Eberhard Backeshoff, DK8. JV. Includes C source code. Also requires VGA video with at least 5. K memory. Shareware, reduced function version of a commercial program from Harlan Technologies. It is a unique system designed for high- level programming on very small microprocessor systems. See the IPS Programming page for more information. IPSDOS. IPS environment emulator for the PC, English version (2. K) and German version (2. K). Includes source code in Pascal. Written by Paul Willmott, VP9. MU. It lets the host computer do almost everything, and is used extensively by amateur satellite groundstations to capture binary telemetry and to implement special broadcast protocols. These programs work with raw binary log files captured from a KISS TNC. KISS to ASCII Converter (1. K). Converts an AX. KISS log file to readable ASCII text, similar to the format used by the monitor mode in some TNCs. Includes C source code. Written by Paul Williamson, KB5. MU. See also the KISS2. ASC web page. You specify what AX. Written by Paul Williamson, KB5. MU. See also the KISSFILT web page. These programs are for use with these spacecraft. See also the Pacsat programs. DOVE OSCAR- 1. 7 Telemetry Decoding Program (2. K). A simple and easy- to- use telemetry decoding program designed to decode ASCII telemetry from DOVE. Version 1. 1 corrects errors in decoding channels 3. Written by John Magliacane, KD2. BD. This one can summarize the history of a particular telemetry channel, so you can see how it has been changing. Documentation in Italian and English. Written by Maurizio Bertolino. Reads binary telemetry from AO- 1. WO- 1. 8, and LO- 1. KISS log file or live from a KISS TNC on COM1 or COM2, and displays all the telemetry channels in familiar engineering units. Can also save the captured telemetry into a KISS log file. Written by Bob Mc. Gwier, N4. HY. Modified by Bruce Rahn, WB9. ANQ. Includes the following utilities. Renames all your KISS log files to match the official naming convention for the telemetry archives. Written by Alberto, I2. KBD. Written by Gerard Regnard, F1. EBN. Written by Harold Price, NK6. K, and Jeff Ward, G0. SUL. Includes C source code and a specification of the BL file format. Written by Jeff Ward, G0. SUL. Includes C source and a specification of the ELTLOG file format. Written by Jeff Ward, G0. SUL. Written by Jeff Ward, G0. SUL. Written by Andre Marchandise, ON1. AIG. Written by Karl- Heinz Muller, VK2. PK. Written by Jeff Ward, G0. SUL. Sat. Sked calculates AOS/LOS times for multiple satellites, has a real time textual position display of up to 2. It conforms to the Kansas City Tracker software specification, and will directly control the KCT board, and the TAPR trakbox. This version of Sat. Sked is Y2. K compatible and no longer requires a registration code to operate. Written by Wayne Roth, WA2. N. Written by Pedro Converso, LU7. ABF. Also provides a simulation mode with which the user can demonstrate many of the Sat. Link functions without actually receiving satellite transmissions. Source code in C (2. K) is also available. See also the author's description (5. K). Written by Jesse Buckwalter, NZ3. F. Written by Chris Jackson, ZL2. TPO. Also uploads and downloads memories. Written by John Wilson, KN4. HX. Includes source code in C++. Written by Anthony Monteiro, AA2. TX, and Howard Long, G6. LVB. The new features include 4x faster tuning. PL encoder control, hot keys to. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
January 2017
Categories |
 RSS Feed
RSS Feed
